


When we hear the word minerals, many may think of dietary supplements or something found in rocks and mountains. But what are minerals really? Minerals are naturally occurring, solid substances with a specific chemical composition and a characteristic, ordered atomic structure. They are the building blocks of many of Earth's rocks and play a crucial role in both nature and technology. In this article, you will get an introduction to the structure, properties, and significance of minerals.
Minerals form when atoms arrange themselves in a regular way and create a crystal lattice. This means that minerals often have characteristic shapes and surfaces. To be considered a mineral, the substance must meet four main requirements:
For example, ice is a mineral (when it occurs naturally, as snow or frost), while glass is not, because glass lacks an ordered structure.
Minerals consist of crystals – that is, their atoms are arranged in a specific pattern that repeats in three dimensions. This is called a crystal lattice. Crystals can take many forms, but the most common crystal structures are divided into seven so-called crystal systems, such as cubic, tetragonal, and hexagonal. Salt (halite) and diamond are both examples of minerals with a well-defined crystal structure.
Each mineral has its own chemical formula. For example, quartz consists of silicon and oxygen (SiO2), while pyrite ("fool's gold") consists of iron and sulfur (FeS2). The composition determines which properties the mineral has – such as color, hardness, and electrical conductivity.
Minerals can be recognized by a number of physical properties, which make them unique and easy to identify:
Color may be the first thing you notice, but it can vary greatly for the same mineral. The streak – the color of the mineral's powder when scratched on a porcelain plate – is often more reliable. For example, hematite is black-gray as a crystal, but has a reddish streak.
Luster describes how the mineral's surface reflects light. It can be metallic, glassy, greasy, or dull. Transparency tells whether light can pass through the mineral – some are transparent (quartz), others completely opaque (galena).
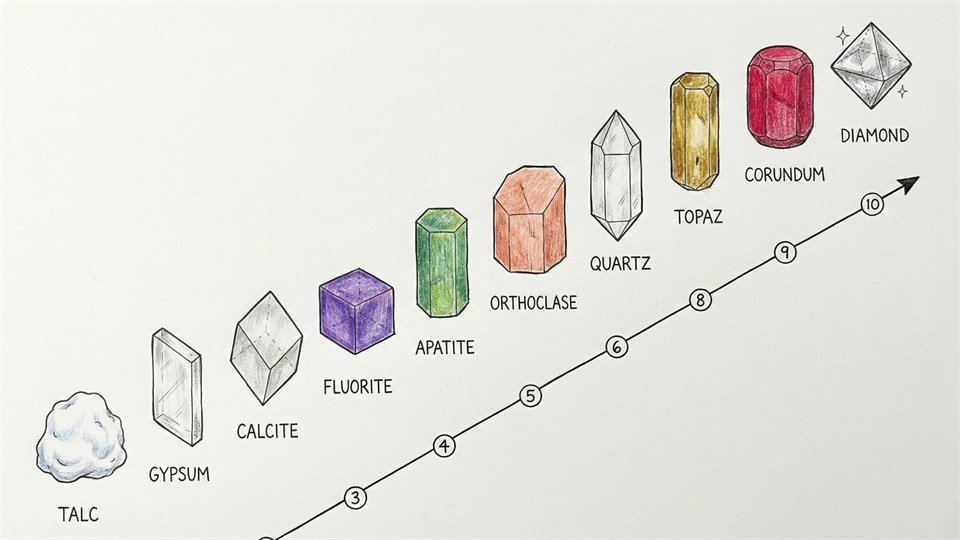
The hardness of minerals is measured according to the Mohs scale of hardness, which goes from 1 (very soft, like talc) to 10 (very hard, like diamond). Hardness determines how easily the mineral can be scratched – and is often used to identify unknown minerals.
Cleavage describes whether the mineral naturally splits along flat surfaces. For example, mica has perfect cleavage and forms thin layers, while quartz does not cleave but breaks into uneven pieces (conchoidal fracture).
Density tells how heavy the mineral feels relative to its size. Magnetism is seen only in a few minerals (such as magnetite), but can be an important property in certain contexts.
Minerals are found everywhere on Earth – in mountains, sand, soil, and even in our bodies. Most rocks consist of a mixture of different minerals. For example, granite consists mainly of quartz, feldspar, and mica. Some minerals can form under specific conditions, such as high pressure or in connection with volcanic activity.
We use minerals every day – often without thinking about it. For example:
Minerals are also found in our bodies – for example, calcium in bones or iron in blood.
Minerals can form in several ways:
These processes take place both deep within the Earth and at the surface, and they make minerals very different in appearance and properties.
Minerals are not only interesting for geologists. They are crucial for modern technology and everyday products. Examples:
Without minerals, our modern society would not function at all. The extraction of minerals (mining) is therefore an important industry, but it also has environmental consequences that require consideration.
The demand for certain minerals is constantly growing – especially for electronics and green energy. This means we must consider how we extract and recycle minerals. Recycling metals and developing environmentally friendly mining methods are central topics for the future if we are to ensure that mineral resources last.
Minerals are small but indispensable building blocks for both nature and our everyday lives. Their structure and unique properties make them the key behind everything from rocks and landscapes to mobile phones and cooking. By understanding the world of minerals, we can better appreciate and protect the resources our planet provides.










