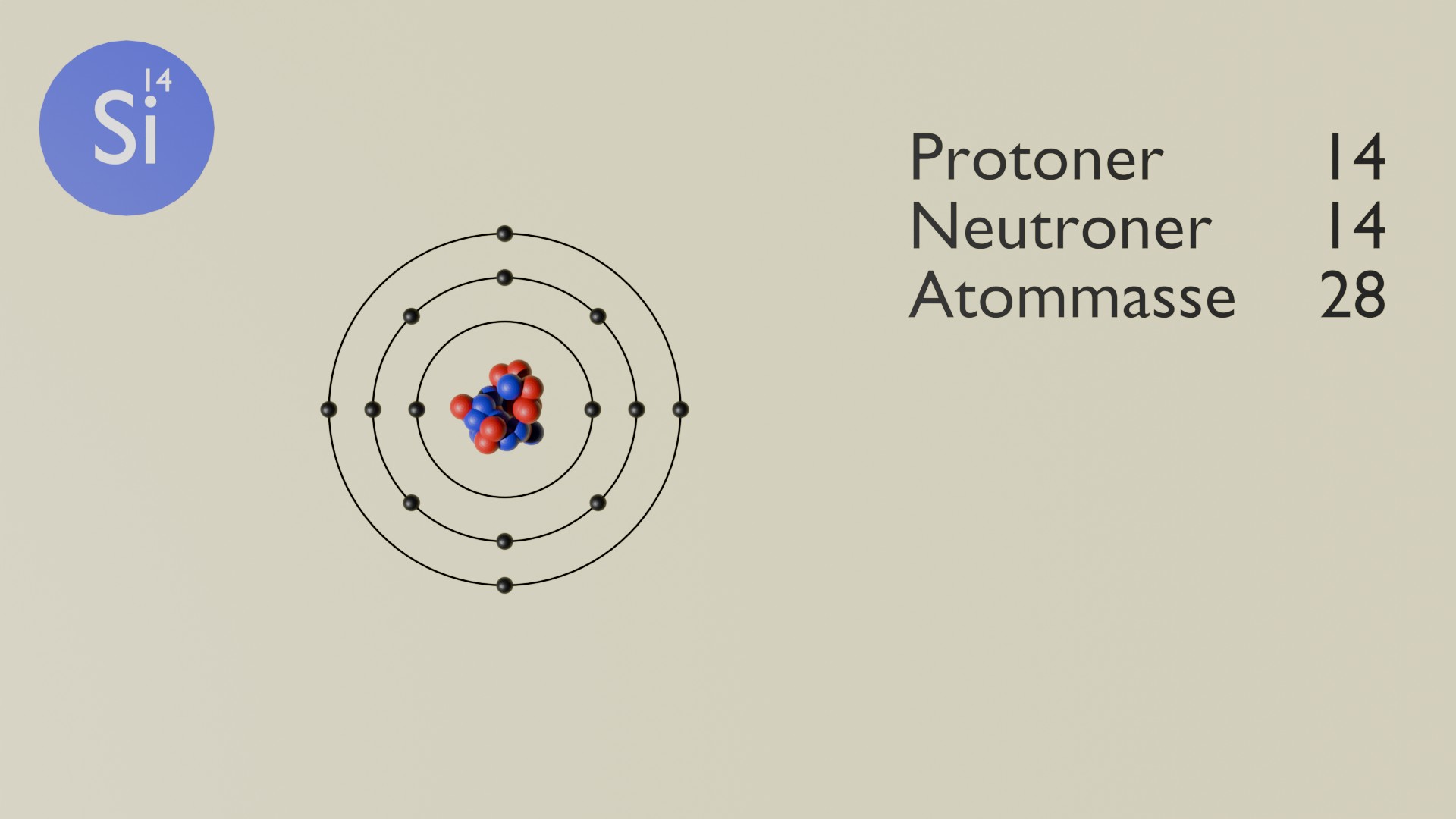
Silicon (Si) is a chemical element with atomic number 14. It is a metalloid and belongs to group 14 in the periodic table. Silicon is the second most abundant element in the Earth's crust and is fundamental to both technology and geology.
1. Introduction to silicon
Silicon has 14 protons, typically 14 neutrons, and 14 electrons. It does not occur freely in nature, but is found in abundant amounts as silicates and quartz. It plays a key role in the manufacture of electronics and glass as well as in biological and geological processes.
2. Physical and chemical properties
Silicon is a grayish, shiny metalloid, which is brittle and has a crystalline structure. It behaves as a non-conductor at low temperatures, but as a semiconductor at higher temperatures – which makes it ideal for electronics.
- Melting point: 1,414 °C
- Boiling point: 3,265 °C
- Semiconductor – electrical conductivity can be controlled
Chemically, silicon is relatively stable, but it can react with halogens and strong bases.
3. Isotopes of silicon
Natural silicon consists of three stable isotopes:
- Silicon-28 (²⁸Si): approx. 92.2 %
- Silicon-29 (²⁹Si): approx. 4.7 %
- Silicon-30 (³⁰Si): approx. 3.1 %
All isotopes are stable and are used in research and advanced material analyses.
4. Industrial applications
Silicon is one of the most widely used elements in modern technology and industry:
| Application | Description |
| Electronics and solar cells | Used in semiconductor chips, transistors, and solar panels due to its controllable conductivity. |
| Building materials | Occurs in cement, concrete, and glass in the form of silicon dioxide (SiO₂). |
| Ceramics and optics | Included in heat-resistant and optical materials such as quartz and silicate glass. |
| Alloys | Silicon is added to aluminum alloys to improve casting properties. |
5. Biological significance and occurrence
Silicon is found in the Earth's crust as silicates and quartz and makes up about 27 % of its mass. It is not essential for humans, but plays a certain role in connective tissue and bones. In plants and certain marine organisms (diatoms), silicon is involved in the formation of cell walls and skeletal structures.