


The periodic table is a tabular arrangement of the chemical elements, organized based on their atomic number, electron configurations, and chemical properties. The elements are arranged in rows (called periods) and columns (called groups or families) in order of increasing atomic number.
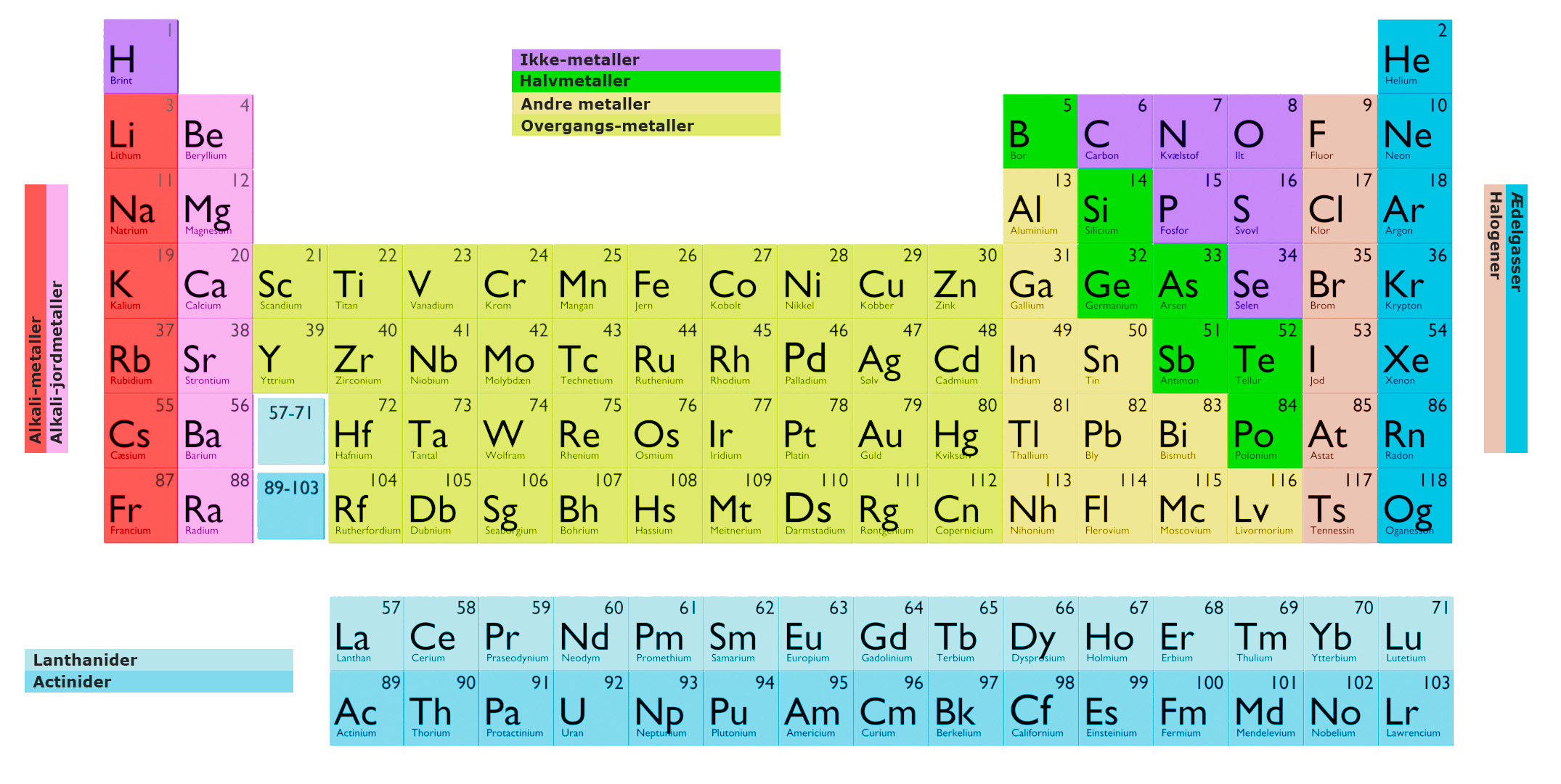
Each element is represented by a unique chemical symbol, typically one or two letters, which is used to identify it in the periodic table. The elements are also grouped into categories based on their physical and chemical properties.
The elements in the first column (called group 1) are called alkali metals and have similar properties. The elements in the second column (group 2) are called alkaline earth metals and have similar properties. The elements in the next six columns (groups 3-8) are called transition metals and have similar properties. The elements in the last column (group 18) are called noble gases and have similar properties.
The rows in the periodic table are called periods. The first row (period 1) has only two elements, hydrogen and helium. The next rows have 8 elements each, from period 2 to period 7.
The elements in the same period have similar electron configurations, but as you move from left to right across a period, the atomic number increases, which leads to an increase in the number of protons in the nucleus, which in turn leads to an increase in the number of electrons in the outermost shell, resulting in different chemical and physical properties.
The elements in the same group (or column) have similar chemical properties because they have the same number of electrons in their outermost shell.
The periodic table is an effective tool for predicting the properties and behavior of elements and their compounds, and it is widely used in chemistry and other sciences.
The alkali metals are a group of elements in the periodic table that are located in group 1 (IA). They are characterized by having one valence electron in their outermost shell. They have similar chemical and physical properties, and they are very reactive. The alkali metals are:
| Element | Description |
|---|---|
| Lithium (Li) | The lightest alkali metal and the least reactive. It is used in batteries and other applications. |
| Sodium (Na) | The most common alkali metal, used in many industrial processes, including the production of glass, soap, and chemicals |
| Potassium (K) | Used in fertilizers, fireworks, and other applications |
| Rubidium (Rb) | Less common and used in specialized applications, such as atomic clocks and research |
| Cesium (Cs) | Is also not commonly used |
| Francium (Fr) | The rarest and most unstable alkali metal and is found only in small amounts in nature |
The alkaline earth metals are a group of elements in the periodic table that are located in group 2 (IIA). They are characterized by having two valence electrons in their outermost shell. They have similar chemical and physical properties, and they are less reactive than the alkali metals. The alkaline earth metals consist of:
| Element | Description |
|---|---|
| Beryllium (Be) | A light and strong metal with unique properties that make it useful in aerospace, nuclear power, and other high-tech applications |
| Magnesium (Mg) | Used in alloys, fireworks, and as a cathode in batteries |
| Calcium (Ca) | An essential element for living organisms and the most abundant metal in the human body. It is used in the construction industry for the production of cement and lime |
| Strontium (Sr) | Used in flares, fireworks, and as a source of strontium-90, a radioactive isotope used in medical treatments. |
| Barium (Ba) | Used in the oil and gas industry, and in the production of glass and ceramics |
| Radium (Ra) | Radioactive and it is only found in small amounts in nature and it has no commercial application |
The transition metals are a group of elements in the periodic table that are located in groups 3-12 (IIIB-IIB) and include the lanthanide and actinide series. They are characterized by having partially filled d-orbitals in their outermost shell.
They have various chemical and physical properties and are known for their high melting and boiling points as well as high densities. They are also known for their ability to form multiple oxidation states and for their colored compounds. Some of the common transition metals are:
| Element | Description |
|---|---|
| Scandium (Sc) | |
| Vanadium (V) | |
| Chromium (Cr) | |
| Manganese (Mn) | |
| Iron (Fe) | |
| Cobalt (Co) | |
| Nickel (Ni) | |
| Copper (Cu) | |
| Zinc (Zn) | |
| Molybdenum (Mo) | |
| Ruthenium (Ru) | |
| Rhodium (Rh) | |
| Palladium (Pd) | |
| Silver (Ag) | |
| Cadmium (Cd) | |
| Mercury (Hg) | |
| Platinum (Pt) | |
| Gold (Au) |
| Element | Description |
|---|---|
| Helium (He) | The second lightest element and used in balloons, airships, and as a coolant for cryogenics |
| Neon (Ne) | Used in neon signs, TV tubes, and as a coolant for cryogenics |
| Argon (Ar) | Used in light bulbs, welding, and as an inert gas in laboratories |
| Krypton (Kr) | Used in flash lamps, strobe lights, and as a coolant for cryogenics |
| Xenon (Xe) | Like krypton, used in flash lamps, strobe lights, and as a coolant for cryogenics |
| Radon (Rn) | Is a radioactive gas and is not used commercially |
These gases are considered chemically inert, which means that they do not participate in chemical reactions, and therefore do not form compounds. However, they can be forced to react under high pressure or extreme temperatures.
